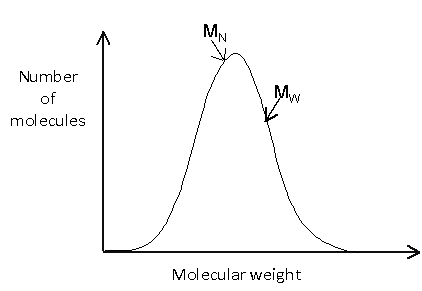
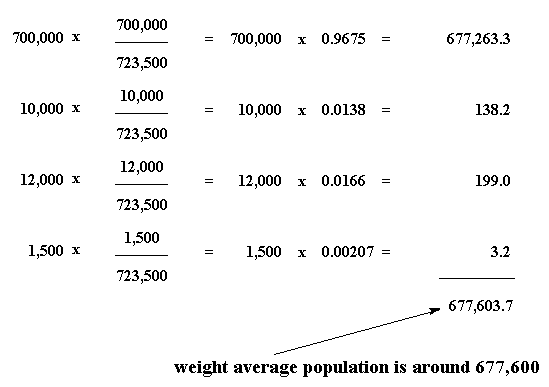
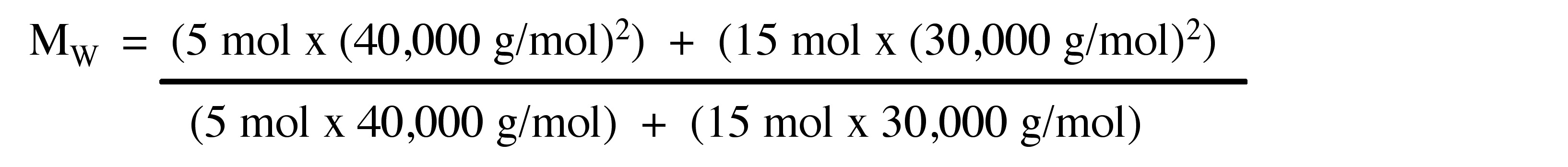A polymeric sample in which 30% molecules have a molecular mass 20,000 , 40% have 30,000 and the rest 30% have 60,000 . The (M̅n) and (M̅w) of this sample are, respectively:
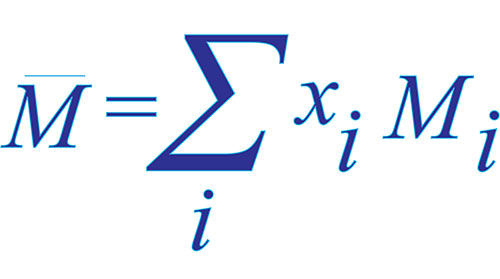
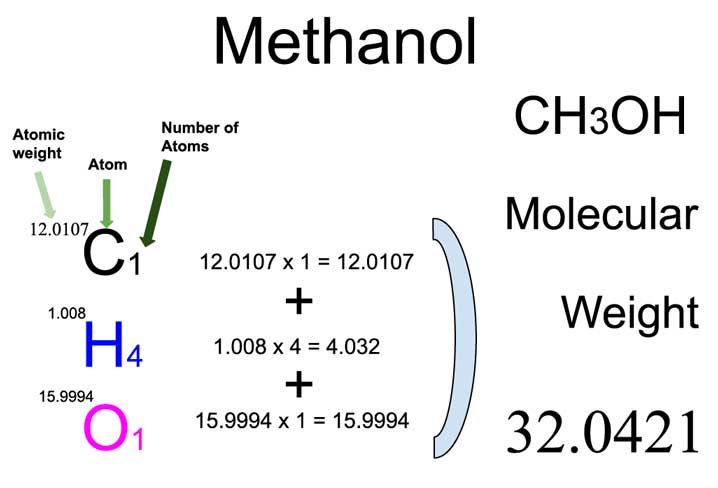
Commercial Hydroponic Farming | How to calculate nutrient content with atomic weight and molecular weight